PDA:PDA质量文化评估工具作为质量管理成熟度推进基础的评估研究
Abstract摘要
The U.S. Food and Drug Administration (FDA) introduced the Quality Management Maturity (QMM) initiative to promote quality practices in pharmaceutical manufacturing that extend beyond basic good manufacturing practices (GMP), as a response to the ongoing drug shortage crisis. As the FDA has yet to formalize a QMM assessment tool, this study evaluated the Parenteral Drug Association’s (PDA) Quality Culture Assessment Tool (QCAT) as a potential surrogate framework. Using anonymized data from 58 life sciences companies, we assessed internal consistency and examined whether the QCAT constructs differentiated responses across organizational, regional, and manufacturing modalities characteristics. Four of five domains—Employee Ownership and Engagement, Continuous Improvement, Technical Excellence, and Communication & Collaboration—demonstrated acceptable internal consistency (Cronbach’s α≥0.70), whereas Leadership Commitment yielded a lower reliability coefficient (α=0.49). Technical Excellence scores significantly differed by business function (p=0.02), with higher ratings reported by innovator firms compared to contract manufacturing organizations (CMO). These findings suggest that the PDA QCAT offers a promising model for operationalizing QMM principles, although refinement may be needed to enhance measurement of leadership constructs.
美国食品药品监督管理局(FDA)推出了质量管理成熟度(Quality Management Maturity, QMM)倡议,旨在促进药品生产中的质量实践超越基础良好生产规范(Good Manufacturing Practices, GMP),以应对持续的药品短缺危机。由于FDA尚未正式制定QMM评估工具,本研究评估了注射剂协会(Parenteral Drug Association, PDA)的质量文化评估工具(Quality Culture Assessment Tool, QCAT)作为潜在的替代框架。研究采用来自58家生命科学公司的匿名化数据,评估了内部一致性,并检验了QCAT结构是否能够区分不同组织、区域和生产模式特征下的响应。五个领域中的四个——员工主人翁意识与参与度(Employee Ownership and Engagement)、持续改进(Continuous Improvement)、技术卓越(Technical Excellence)和沟通与协作(Communication & Collaboration)——表现出可接受的内部一致性(克朗巴赫系数Cronbach’s α≥0.70),而领导层承诺(Leadership Commitment)则显示出较低的可靠性系数(α=0.49)。技术卓越得分因业务功能不同而存在显著差异(p=0.02),创新型企业报告的评分高于合同生产组织(Contract Manufacturing Organizations, CMO)。这些发现表明,PDA QCAT为QMM原则的操作化提供了一个有前景的模型,尽管可能需要进一步完善以增强领导力构念的测量。
Keywords: leadership commitment · parenteral drug association’s (PDA) quality culture assessment tool (QCAT) · pharmaceutical quality systems · quality culture · quality management maturity (QMM) · technical excellence
关键词: 领导承诺 · 注射剂协会(PDA)质量文化评估工具(QCAT) · 药品质量体系 · 质量文化 · 质量管理成熟度(QMM) · 技术卓越
Introduction介绍
The U.S. Food and Drug Administration (FDA) established the Quality Management Maturity (QMM) initiative as a strategic framework to elevate pharmaceutical manufacturing standards beyond baseline compliance with Current Good Manufacturing Practices (cGMP). Launched in response to persistent drug shortages, QMM promotes proactive quality oversight and aims to strengthen supply chain resilience, foster a culture of continuous improvement, and integrate quality management as a strategic advantage rather than a regulatory obligation (1).
美国食品药品监督管理局(FDA)将质量管理成熟度(Quality Management Maturity, QMM)倡议确立为一项战略框架,旨在将药品生产标准提升至超越现行良好生产规范(Current Good Manufacturing Practices, cGMP)基础合规水平。该倡议针对持续的药品短缺问题而启动,倡导主动质量监管,旨在增强供应链韧性,培育持续改进文化,并将质量管理整合为战略优势而非仅仅是监管义务(1)。
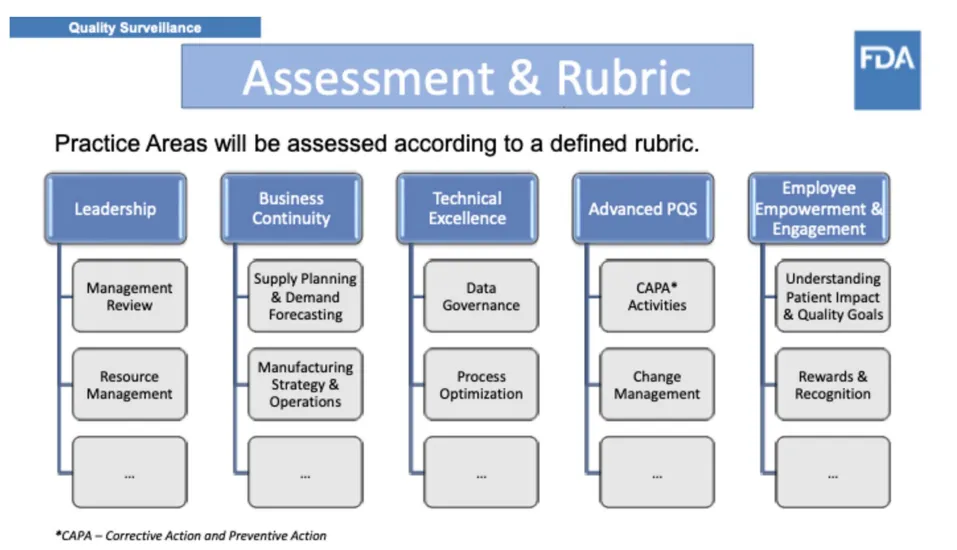
While QMM remains under development, the FDA has articulated its broad goals and identified five practice areas, which are intended to serve as the program’s foundational domains and are displayed in Fig. 1: Leadership, Business Continuity, Technical Excellence, Advanced Pharmaceutical Quality Systems (PQS), and Employee Empowerment and Engagement (2–4). Notably, QMM differs in scope and intent from FDA’s Quality Metrics program, which quantifies operational data, and from existing industry-driven quality culture assessments (5, 6). QMM seeks to evaluate the maturity of the systems, behaviors, and leadership practices that underpin consistent, high-quality pharmaceutical manufacturing.
尽管QMM仍在开发中,FDA已阐明其广泛目标并确定了五个实践领域,这些领域拟作为该计划的基础领域,如图1所示:领导力(Leadership)、业务连续性(Business Continuity)、技术卓越(Technical Excellence)、先进药品质量体系(Advanced Pharmaceutical Quality Systems, PQS)以及员工赋能与参与(Employee Empowerment and Engagement)(2-4)。值得注意的是,QMM在范围和意图上不同于FDA的质量指标(Quality Metrics)计划——后者量化运营数据,也不同于现有的行业主导的质量文化评估(5,6)。QMM旨在评估支撑一致、高质量药品生产的体系、行为和领导实践的成熟度。
While achieving widespread support for the QMM framework, key questions remain regarding its implementation, standardization, and feasibility across diverse manufacturing contexts. Stakeholders have voiced concerns about potential burdens on small and medium-sized firms, the risks of public score disclosure, and the need for validated and actionable metrics (7–9). Nevertheless, many manufacturers recognize the program’s potential value in regulatory interactions, in enhancing reputational standing, and in minimizing the risk of supply disruptions (10). Emerging industry sentiment suggests that healthcare purchasers and payers may accept modest price premiums in exchange for higher assurance of supply continuity, especially in high-risk therapeutic areas (11).
尽管QMM框架获得了广泛支持,但关于其在不同生产背景下的实施、标准化和可行性仍存在关键问题。利益相关方已表达对中小型企业的潜在负担、公开评分披露的风险以及对经验证且可操作的指标的需求等方面的担忧(7-9)。尽管如此,许多生产商认识到该计划在监管互动、提升声誉地位以及最小化供应中断风险方面的潜在价值(10)。新兴的行业表明,医疗采购方和支付方可能愿意为更高的供应连续性保证接受适度的价格溢价,尤其是在高风险治疗领域(11)。
A strong organizational quality culture is increasingly seen as a cornerstone of QMM (3–5, 10–17). As emphasized in FDA guidance and white papers, executive commitment, workforce empowerment, and embedded quality values influence not only operational outcomes but also long-term reliability and regulatory trust (1, 11, 13). Previous research in collaboration with the Parenteral Drug Association (PDA) supports this position, showing that employee quality behaviors are positively correlated with site-level quality maturity that further ensures consistent drug product quality and sustainable supply (16, 17).
强大的组织质量文化日益被视为QMM的基石(3-5,10-17)。正如FDA指南和白皮书所强调的,高管承诺、员工赋能和根深蒂固的质量价值观不仅影响运营结果,还影响长期可靠性和监管信任(1,11,13)。此前与注射剂协会(PDA)合作的研究支持这一观点,表明员工质量行为与场地级质量成熟度呈正相关,进而确保一致的药品质量和可持续供应(16,17)。
Drug shortages remain a persistent threat to patient care, often resulting from insufficiently mature quality management systems that fail to detect and prevent supply chain vulnerabilities (1, 11). As emphasized in Harouaka et al.‘s [3] FDA QMM Overview, the framework aims to address these deficiencies by promoting data-driven, preventive quality systems that enhance production reliability and transparency (3). Pilot programs conducted by the US FDA CDER from 2020 to 2022 evaluated QMM implementation among U.S.-based finished dosage form (FDF) manufacturers and international active pharmaceutical ingredient (API) producers, offering transparency into the program’s practical application and highlighting areas for refinement (10). Building on these efforts, the Benchmarking of the Quality Practices of Global Pharmaceutical Manufacturing to Advance Supply Chain Resilience report (18) and Harouaka et al. (3) further underscore QMM’s potential to stabilize global supply chains by linking mature quality management practices to improved access and reduced risk of product recalls.
药品短缺仍然对患者护理构成持续威胁,这通常源于不够成熟的质量管理体系未能发现和预防供应链脆弱性(1,11)。正如Harouaka等人在[3]FDA QMM概述中强调的,该框架旨在通过推广数据驱动的预防性质量系统来解决这些缺陷,以增强生产可靠性和透明度(3)。美国FDA药品审评与研究中心(CDER)于2020至2022年开展的试点项目评估了QMM在美国本土制剂成品(Finished Dosage Form, FDF)生产商和国际原料药(Active Pharmaceutical Ingredient, API)生产商中的实施情况,为该计划的实际应用提供了透明度并突出了需要改进的领域(10)。在这些工作的基础上,《全球药品生产质量实践基准测试以促进供应链韧性》报告(18)和Harouaka等人(3)进一步强调了QMM通过将成熟的质量管理实践与改善药品可及性和降低产品召回风险相联系,从而稳定全球供应链的潜力。
Our study evaluates the PDA’s Quality Culture Assessment Tool (QCAT) as a potential operational model to support the QMM initiative. Our goal is to determine whether QCAT offers a valid, data-driven foundation for measuring the types of quality maturity behaviors that QMM aims to promote.
本研究评估了PDA质量文化评估工具(QCAT)作为支持QMM倡议的潜在操作模型。我们的目标是确定QCAT是否提供了一个有效的、数据驱动的基础,用于测量QMM旨在推广的质量成熟度行为类型。
Materials and Methods
材料与方法
Study Objective
研究目标
This study evaluates quality culture scores submitted anonymously to the Parenteral Drug Association (PDA) by multiple life sciences companies. By analyzing these data, we aimed to identify trends in quality culture practices and assess the suitability of the QCAT tool as a benchmarking instrument for organizational maturity. The tool utilized a survey interview approach to evaluate quality culture at the manufacturing site level.
本研究评估了多家生命科学公司匿名提交给注射剂协会(PDA)的质量文化评分。通过分析这些数据,我们旨在识别质量文化实践的趋势,并评估QCAT工具作为组织成熟度基准测试工具的适用性。该工具采用调查访谈方法来评估生产场地级别的质量文化。
Survey Administration and Dataset Composition
调查实施与数据集构成
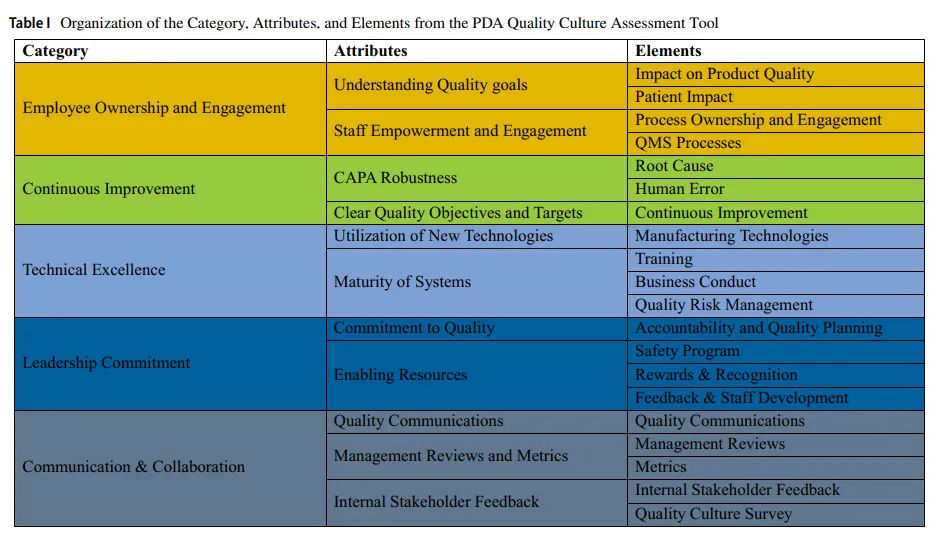
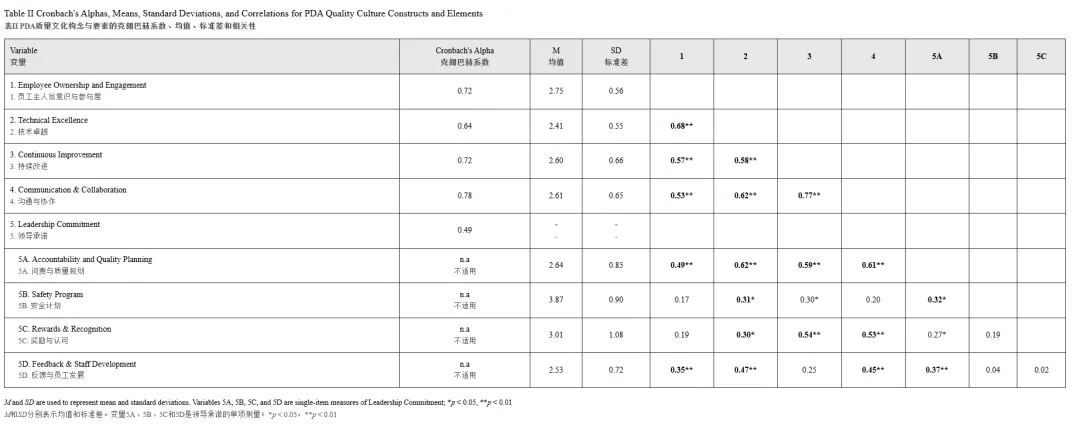
The PDA QCAT tool consists of 20 elements, each representing a specific behavioral or operational practice. These elements are organized into 11 attributes, which in turn roll up into 5 overarching categories as follows (also shown in Table I): 1: Employee Ownership and Engagement, 2: Continuous Improvement, 3: Technical Excellence, 4: Leadership Commitment, and 5: Communication & Collaboration.
PDA QCAT工具包含20个要素(elements),每个要素代表特定的行为或操作实践。这些要素被组织成11个属性(attributes),进而汇总为5个 overarching 类别(categories),如下所示(亦见表I):1:员工主人翁意识与参与度(Employee Ownership and Engagement)、2:持续改进(Continuous Improvement)、3:技术卓越(Technical Excellence)、4:领导承诺(Leadership Commitment)、5:沟通与协作(Communication & Collaboration)。
Each element is scored on a five-point maturity scale, where one (1) represents the least mature state and five (5) the most mature expression of the element. While suggested probing questions and rating anchors are provided to guide evaluation, the determination of scores is ultimately left to the discretion of PDA trained assessors based on structured interviews and observations.
每个要素采用五点成熟度量表评分,其中1分代表最不成熟状态,5分代表该要素最成熟的表达。虽然提供了建议的探查问题和评分锚点以指导评估,但最终评分由经PDA培训的评估员基于结构化访谈和观察酌情决定。
PDA-trained internal employees conducted the site-level assessments using the QCAT framework. After completing the training, each assessor returned to their organization and independently evaluated one manufacturing site, scoring all 20 elements based on structured interviews and site observations. Participating companies could submit their raw data to PDA for benchmarking. PDA anonymized all submissions, and no company-identifying information was retained. Element scores were submitted as whole numbers and not aggregated or normalized. This study analyzed only the raw element-level scores.
经PDA培训的内部员工使用QCAT框架进行场地级评估。完成培训后,每位评估员返回其组织并独立评估一个生产场地,基于结构化访谈和场地观察对所有20个要素进行评分。参与公司可将其原始数据提交给PDA进行基准测试。PDA对所有提交数据进行匿名化处理,不保留任何可识别公司的信息。要素评分以整数提交,未进行汇总或标准化处理。本研究仅分析原始要素级评分。
A total of 58 life sciences companies participated in the PDA QCAT assessment and submitted their results. PDA anonymized all submissions before analysis. Participating companies represent a wide cross-section of the life science industry, encompassing a variety of modalities and operational roles.
共有58家生命科学公司参与PDA QCAT评估并提交了结果。PDA在分析前对所有提交数据进行了匿名化处理。参与公司代表了生命科学行业的广泛横截面,涵盖多种生产模式和运营角色。
Survey respondents identified the characteristics of their manufacturing sites across several organizational dimensions. These included geographic location (North America, Europe, and Asia); business function (e.g., innovator company, contract manufacturing organization, generic manufacturer, over-the-counter producer); product class (small molecule, biologics/biotech, vaccines); product type (non-sterile finished product, sterile finished product, or active pharmaceutical ingredient/drug substance); and employee size (categorized as fewer than 1,000, greater than 1,000, or not specified). This level of segmentation enabled comparative analysis across multiple operational and demographic variables.
调查受访者从多个组织维度识别了其生产场地的特征。这些维度包括地理位置(北美、欧洲和亚洲);业务功能(如创新公司、合同生产组织、仿制药生产商、非处方药生产商);产品类别(小分子、生物制品/生物技术、疫苗);产品类型(非无菌成品、无菌成品或原料药/药物物质);以及员工规模(分类为少于1,000人、大于1,000人或未指定)。这种细分水平实现了跨多个运营和人口统计变量的比较分析。
Because PDA offers QCAT training and benchmarking on a rolling basis, the dataset reflects an evolving snapshot of quality culture maturity across the global pharmaceutical industry. While this study offers a cross-sectional view, the framework supports future longitudinal tracking.
由于PDA以滚动方式提供QCAT培训和基准测试,该数据集反映了全球制药行业质量文化成熟度的动态快照。虽然本研究提供横断面视角,但该框架支持未来的纵向追踪。
Analysis and Results
分析与结果
To examine the interrelationships among the elements of pharmaceutical quality culture, we computed bivariate correlations among individual quality culture elements. As shown in Supplementary Table 1, the results revealed a high level of interdependence within the quality culture framework, with many elements exhibiting significant associations and only a few showing nonsignificant relationships.
为检验药品质量文化要素之间的相互关系,我们计算了各质量文化要素间的双变量相关性。如补充表1所示,结果显示质量文化框架内存在高水平的相互依赖性,许多要素表现出显著相关性,仅少数显示无显著关系。
To explore convergent and divergent validity, we examined Pearson correlations among the 20 QCAT elements (see Supplementary Table S1). As anticipated, elements grouped within the same conceptual category tended to exhibit stronger positive associations. The strongest observed correlation was between Management Review and Metrics (r=0.75, p<0.01), both belonging to the Communication & Collaboration domain. Similarly, Internal Stakeholder Feedback and Quality Culture Survey, also within the same category, showed a robust correlation (r=0.61, p<0.01). Notably, the most pronounced cross-domain relationship emerged between Continuous Improvement (from the Continuous Improvement category) and Quality Communications (from Communication & Collaboration), which also correlated strongly (r=0.65, p<0.01). This suggests that organizations emphasizing process improvement often also demonstrate strong communication practices, highlighting the cross-functional nature of quality culture development.
为探索聚合效度和区分效度,我们检验了20个QCAT要素之间的皮尔逊相关性(见补充表S1)。正如预期,同一概念类别内的要素往往表现出更强的正相关。观察到的最强相关性存在于管理评审与指标(Management Review and Metrics)之间(r=0.75, p<0.01),二者均属于沟通与协作(Communication & Collaboration)领域。同样,内部利益相关方反馈与质量文化调查(Internal Stakeholder Feedback and Quality Culture Survey)也在同一类别内显示出稳健的相关性(r=0.61, p<0.01)。值得注意的是,最显著的跨领域关系出现在持续改进(来自持续改进类别)与质量沟通(来自沟通与协作)之间,二者也高度相关(r=0.65, p<0.01)。这表明强调过程改进的组织通常也表现出强大的沟通实践,凸显了质量文化发展的跨职能性质。
Several cross-domain elements showed no meaningful association. For instance, Impact on Product Quality and Rewards and Recognition were uncorrelated (r=−0.001, ns), pointing to a disconnect between recognition systems and perceived product quality outcomes. Similarly, Internal Stakeholder Feedback and Manufacturing Technologies (r=0.004, ns) showed no relationship, possibly reflecting the organizational separation between engagement and technical infrastructure. Even within a single category, some elements exhibited limited coherence. Although grouped under Leadership Commitment category, Feedback and Staff Development element showed negligible associations with both Safety Program (r=0.04, ns) and Rewards and Recognition (r=0.04, ns). These findings raise concerns about the conceptual coherence and internal consistency of this category.
若干跨领域要素未显示出有意义的相关性。例如,对产品质量的影响(Impact on Product Quality)与奖励与认可(Rewards and Recognition)无相关性(r=-0.001, ns),表明认可体系与感知的产品质量结果之间存在脱节。同样,内部利益相关方反馈(Internal Stakeholder Feedback)与生产技术(Manufacturing Technologies)(r=0.004, ns)无相关性,可能反映了参与度与技术基础设施之间的组织分离。即使在单一类别内,某些要素也表现出有限的连贯性。虽然被归入领导承诺(Leadership Commitment)类别,但反馈与员工发展(Feedback and Staff Development)要素与安全计划(Safety Program)(r=0.04, ns)和奖励与认可(r=0.04, ns)均显示出可忽略的相关性。这些发现引发了关于该类别的概念连贯性和内部一致性的担忧。
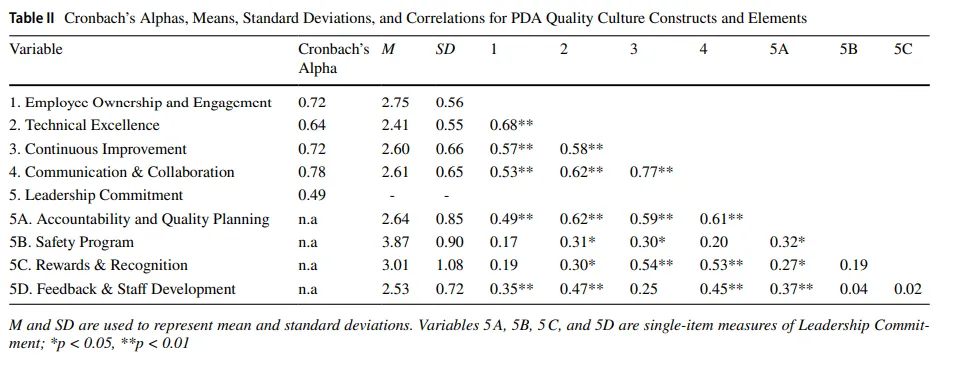
To evaluate the structural and functional robustness of the PDA QCAT tool, we conducted a two-pronged analysis assessing internal consistency and between-group sensitivity across a sample of 58 life sciences organizations. First, we examined internal consistency and reliability across the tool’s five categories: Employee Ownership and Engagement, Continuous Improvement, Technical Excellence, Leadership Commitment, and Communication & Collaboration. Cronbach’s alpha values, summarized in Table II, indicated acceptable internal consistency for four of the five categories, with Leadership Commitment being the only exception. This pattern was broadly consistent with expectations, as the PDA framework groups conceptually aligned elements within each domain; however, the comparatively low reliability of the Leadership Commitment category suggests that its elements may not function as a unified construct in the same manner as the other domains. Because Leadership Commitment demonstrated low internal consistency (Cronbach’s α=0.49), we proceeded with its four elements individually rather than as a composite score. While originally conceptualized as a unified domain, the observed inconsistency led us to interpret Leadership Commitment descriptively rather than as a psychometrically robust construct. Accordingly, these elements were retained for exploratory purposes and analyzed at the item level across subsequent comparisons.
为评估PDA QCAT工具的结构和功能稳健性,我们对58家生命科学组织样本进行了双管齐下的分析,评估内部一致性和组间敏感性。首先,我们检验了工具五个类别的内部一致性和可靠性:员工主人翁意识与参与度、持续改进、技术卓越、领导承诺和沟通与协作。表II总结的克朗巴赫系数(Cronbach’s alpha)值显示,五个类别中有四个具有可接受的内部一致性,领导承诺是唯一例外。这一模式与预期基本一致,因为PDA框架在每个领域内对概念上对齐的要素进行分组;然而,领导承诺类别相对较低的可靠性表明其要素可能不像其他领域那样作为统一构念发挥作用。由于领导承诺显示出较低的内部一致性(克朗巴赫系数α=0.49),我们对它的四个要素进行单独处理而非作为综合评分。虽然最初被概念化为统一领域,但观察到的非一致性使我们倾向于对领导承诺进行描述性解释,而非作为心理测量学上稳健的构念。因此,这些要素被保留用于探索性目的,并在后续比较中进行项目水平分析。
To examine the structural coherence of the PDA QCAT tool, we calculated Pearson correlations among the four reliable composite constructs and the individual items under Leadership Commitment—namely, Accountability and Quality Planning, Safety Program, Rewards and Recognition, and Feedback and Staff Development. The four composites demonstrated strong and statistically significant intercorrelations (ranging from r=0.53 to r=0.77, all p<0.01, as shown in Table II), indicating a high degree of internal structure and shared variance among the dimensions of quality culture. In contrast, the individual Leadership Commitment elements showed only modest and inconsistent associations with the composite constructs. For instance, Accountability and Quality Planning was significantly correlated with all four composites (r=0.49 to 0.62), whereas Safety Program, Rewards and Recognition, and Feedback and Staff Development lacked consistent alignment and were weakly or not significantly related to other dimensions, nor to other elements within Leadership Commitment category, as described above.
为检验PDA QCAT工具的结构连贯性,我们计算了四个可靠综合构念与领导承诺下各单项之间的皮尔逊相关性——即问责与质量规划(Accountability and Quality Planning)、安全计划、奖励与认可以及反馈与员工发展。四个综合构念显示出强且统计上显著的相互相关性(范围从r=0.53到r=0.77,所有p<0.01,见表II),表明质量文化各维度之间具有高度的内部结构和共享方差。相比之下,领导承诺的各单项与综合构念仅显示出适度且不一致的相关性。例如,问责与质量规划与所有四个综合构念显著相关(r=0.49至0.62),而安全计划、奖励与认可以及反馈与员工发展缺乏一致的对齐,与其他维度或领导承诺类别内其他要素的关联较弱或无显著相关性,如上所述。
Group-Level Differences Across Organizational Characteristics
跨组织特征的组间差异
Composite Categories
综合类别
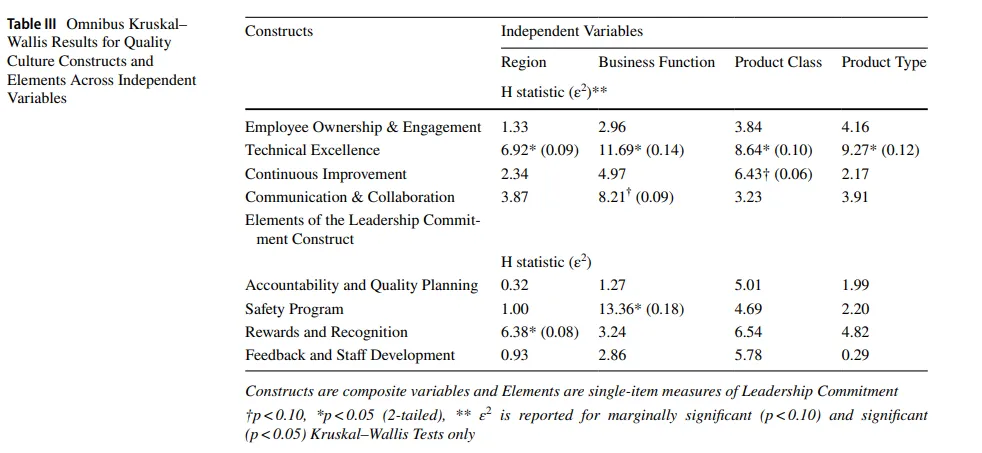
To examine whether quality culture perceptions varied across key organizational characteristics, we conducted a series of Kruskal–Wallis H tests for each of the four internally consistent composite constructs: Employee Ownership and Engagement, Technical Excellence, Continuous Improvement, and Communication & Collaboration. Independent variables included Geographic Region, Business Function, Product Class, and Product Type. Given the ordinal nature of the data and moderate sample sizes, nonparametric methods were used to avoid violations of ANOVA assumptions. Epsilon-squared (ε²) effect sizes were calculated for Kruskal–Wallis tests that yielded statistically significant or marginal p-values (p<0.10). Post hoc pairwise comparisons were conducted using Dunn’s test with Bonferroni correction where Kruskal–Wallis results were significant (p<0.05). A summary of test results is presented in Table III.
为检验质量文化感知是否因关键组织特征而异,我们对四个内部一致的综合性构念分别进行了一系列Kruskal-Wallis H检验:员工主人翁意识与参与度、技术卓越、持续改进和沟通与协作。自变量包括地理区域、业务功能、产品类别和产品类型。鉴于数据的序数性质和中等样本量,使用非参数方法以避免违反方差分析(ANOVA)假设。对产生统计显著或边缘p值(p<0.10)的Kruskal-Wallis检验计算了ε平方(epsilon-squared, ε²)效应量。在Kruskal-Wallis结果显著(p<0.05)时,采用Dunn检验结合Bonferroni校正进行事后两两比较。检验结果汇总于表III。
Scores for Employee Ownership and Engagement, Continuous Improvement, and Communication & Collaboration did not significantly differ across any of the organizational characteristics examined (see Table III), suggesting broadly consistent ratings across contexts. A marginal effect for product class on Continuous Improvement (H(3)=6.43, p=0.09) and for business function on Communication & Collaboration (H(4)=8.21, p=0.08) did not yield significant post hoc differences after Bonferroni correction.
员工主人翁意识与参与度、持续改进和沟通与协作的得分在检验的任何组织特征间均无显著差异(见表III),表明跨背景评分基本一致。产品类别对持续改进的边缘效应(H(3)=6.43, p=0.09)和业务功能对沟通与协作的边缘效应(H(4)=8.21, p=0.08)在Bonferroni校正后未产生显著的事后差异。
Technical Excellence differed significantly across business functions (H(4)=11.69, p=0.02, ε²=0.15).
技术卓越在业务功能间存在显著差异(H(4)=11.69, p=0.02, ε²=0.15)。
Dunn–Bonferroni post hoc tests indicated higher scores for innovator organizations than for CMOs (p=0.01), and this was the only pairwise comparison that remained significant after adjustment (all other adjusted p>0.25). Although omnibus tests for Technical Excellence by region, product class, and product type reached nominal significance (ε²=0.09–0.11), none of their adjusted pairwise comparisons were significant.
Dunn-Bonferroni事后检验显示创新型组织评分高于CMO(p=0.01),这是校正后唯一保持显著的两两比较(所有其他校正后p>0.25)。虽然按区域、产品类别和产品类型对技术卓越进行的总检验达到了名义显著性(ε²=0.09-0.11),但其校正后的两两比较均无显著性。
Leadership Commitment Elements
领导承诺要素
To further evaluate the PDA QCAT tool, we examined group-level differences in the four elements comprising the Leadership Commitment category: Accountability and Quality Planning, Safety Program, Rewards and Recognition, and Feedback and Staff Development. Given the low internal consistency of this category (Cronbach’s α=0.49), each element was analyzed separately using Kruskal–Wallis H tests across geographic region, business function, product class, and product type, summarized in Table III.
为进一步评估PDA QCAT工具,我们检验了领导承诺类别中四个要素的组间差异:问责与质量规划、安全计划、奖励与认可以及反馈与员工发展。鉴于该类别的内部一致性较低(克朗巴赫系数α=0.49),每个要素分别使用Kruskal-Wallis H检验按地理区域、业务功能、产品类别和产品类型进行分析,汇总于表III。
Rewards and Recognition differed across regions (H(2)=6.38, p=0.04, ε²=0.08), with Dunn–Bonferroni tests indicating higher scores in North America than in Asia (adjusted p=0.02). Safety Program varied by business function (H(4)=13.36, p=0.01, ε²=0.18), with generic firms scoring lower than innovator organizations. No other comparisons reached significance. Together, these findings suggest that perceptions of Leadership Commitment are largely stable, but some variation emerges across regions and by business function or employee recognition.
奖励与认可在区域间存在差异(H(2)=6.38, p=0.04, ε²=0.08),Dunn-Bonferroni检验显示北美评分高于亚洲(校正后p=0.02)。安全计划因业务功能而异(H(4)=13.36, p=0.01, ε²=0.18),仿制药公司评分低于创新型组织。其他比较均未达到显著性。总之,这些发现表明领导承诺的感知基本稳定,但在区域间以及按业务功能或员工认可方面出现一些变异。
Discussion
讨论
Taken together, these findings indicate that although the PDA Quality Culture Assessment Tool (QCAT) demonstrates internal coherence and a theoretically consistent domain structure, its ability to discriminate between sites appears limited in the present dataset. The small number of significant post hoc differences, coupled with compressed response distributions for several elements, suggests constrained variability at the site level. This pattern may reflect socially desirable responding, genuine convergence in quality culture across participating firms, or a need for greater scale granularity to enhance benchmarking sensitivity. These considerations are particularly relevant for the potential use of the instrument in the context of Quality Management Maturity (QMM), where meaningful differentiation between sites is a central objective.
总体而言,这些发现表明,尽管PDA质量文化评估工具(QCAT)显示出内部连贯性和理论上一致的领域结构,但在当前数据集中其区分不同场地的能力似乎有限。显著事后差异数量较少,加上若干要素的响应分布压缩,表明场地级变异受限。这一模式可能反映了社会期望性应答、参与公司间质量文化的真正趋同,或需要更精细的量表粒度以增强基准测试敏感性。这些考虑对于该工具在质量管理成熟度(QMM)背景下的潜在使用尤为重要,因为在QMM中场地间的有意义区分是核心目标。
Measuring Leadership Commitment
测量领导承诺
The Leadership Commitment category demonstrated lower internal consistency, suggesting potential limitations in either the construct’s operationalization or the consistency with which organizations interpret and report the associated behaviors. This result highlights the need for further refinement of the underlying measurement model. Specifically, the Leadership Commitment element Accountability and Quality Planning was the sole element demonstrating robust correlation with all composites/categories, implying that it may serve as a more central leadership indicator than the remaining elements. In contrast, the weaker associations of Safety Program and Rewards and Recognition suggest that these elements may not be perceived as leadership-driven behaviors within the context of quality culture. It is also important to note that safety programs in many facilities, especially in the United States, are frequently managed by Environmental Health and Safety functions rather than by quality departmental functions. As a result, inclusion of safety-related metrics within the Leadership Commitment category may introduce construct heterogeneity that contributes to the observed internal inconsistency.
领导承诺类别显示出较低的内部一致性,表明要么该构念的操作化存在潜在局限,要么组织在解释和报告相关行为时的一致性不足。这一结果凸显了对基础测量模型进一步完善的需要。具体而言,领导承诺要素问责与质量规划是唯一与所有综合构念/类别显示出稳健相关性的要素,暗示其可能比其余要素更适合作为核心领导指标。相比之下,安全计划和奖励与认可的较弱相关性表明,在质量文化背景下,这些要素可能不被视为领导驱动的行为。还需注意的是,许多场地中的安全计划,尤其在美国,通常由环境健康与安全(Environmental Health and Safety, EHS)职能管理而非质量部门职能管理。因此,在领导承诺类别中包含安全相关指标可能引入构念异质性,从而导致观察到的内部非一致性。
Contextual Variability in Technical Excellence
技术卓越的情境变异性
The results revealed that QMM cultural dimensions are not uniformly distributed across business models and product types. Technical Excellence scores were significantly higher among facilities producing innovator drugs compared to contract manufacturing organizations (CMOs), potentially reflecting greater investments in proprietary technology and quality infrastructure. Innovator organizations generally have greater access to resources, enabling them to recruit and retain highly specialized talent and support state-of-the-art capabilities. In contrast, CMOs often prioritize operational efficiencies and optimize yields in their investment strategies, given the surmounting pressures of reducing operational costs.
结果显示QMM文化维度在不同商业模式和产品类型间分布不均。技术卓越得分在生产创新药物的场地中显著高于合同生产组织(CMO),这可能反映了对专有技术和质量基础设施的更大投资。创新型组织通常拥有更丰富的资源获取渠道,使其能够招聘和保留高度专业化人才并支持最先进的能力。相比之下,鉴于降低运营成本的不断增加的压力,CMO通常在投资策略中优先考虑运营效率和收率优化。
Implications for QMM Operationalization
对QMM操作化的启示
These findings carry important implications for the FDA’s ongoing efforts to formalize and operationalize the QMM program. The strong intercorrelations observed among key quality culture variables support the inclusion of both human-centric and system-level indicators within any prospective QMM assessment framework. The internal consistency of most QCAT domains suggests that the PDA instrument can serve as a credible foundation for QMM evaluations. Collectively, these results reinforce the centrality of quality culture as a foundational construct within the QMM architecture and highlight the value of empirically grounded frameworks in driving consistent and meaningful maturity assessments across the pharmaceutical manufacturing sector.
这些发现对FDA持续努力正式制定和操作化QMM计划具有重要启示。观察到的关键质量文化变量间的强相互相关性支持在任何前瞻性QMM评估框架中包含以人为中心和系统层面的指标。大多数QCAT领域的内部一致性表明PDA工具可作为QMM评估的可信基础。总体而言,这些结果强化了质量文化作为QMM架构基础构念的核心地位,并凸显了经验证框架在推动制药生产领域一致且有意义的成熟度评估方面的价值。
Importantly, the observed differences in Technical Excellence across business functions (i.e., innovators, CMOs, generics, and OTC manufacturers) highlight the need for contextualized benchmarking within the QMM framework. A one-size-fits-all maturity index may fail to reflect the distinct priorities of different business models (e.g., innovation vs. efficiency). Tailored evaluation frameworks that align with the strategic objectives of each organizational type may improve the utility and fairness of QMM scoring.
重要的是,观察到的技术卓越在业务功能间(即创新型企业、CMO、仿制药企业和非处方药生产商)的差异凸显了在QMM框架内进行情境化基准测试的必要性。一刀切的成熟度指数可能无法反映不同商业模式(如创新与效率)的 distinct 优先事项。与每种组织类型的战略目标相一致的有针对性的评估框架可能提高QMM评分的实用性和公平性。
QCAT’s Comparability
QCAT的可比性
While the QCAT provides a foundational framework for assessing quality culture, it is important to note that it may not fully capture the nuanced rationale underlying individual site scores. To enhance comparability, it is recommended that assessors and respondents be given the opportunity to include structured qualitative justifications for each rating; whether for quality culture or future QMM framework. Such contextual narratives would facilitate a more holistic evaluation by clarifying site-specific considerations, organizational priorities, or operational barriers, and would support more meaningful comparisons across organizations.
虽然QCAT为评估质量文化提供了基础框架,但需要注意的是,它可能无法完全捕捉支撑单个场地评分的细微原理。为增强可比性,建议给予评估员和受访者机会,为每项评分提供结构化的定性理由;无论是针对质量文化还是未来的QMM框架。这种情境化叙述将通过澄清场地特定考虑、组织优先事项或运营障碍来促进更全面的评估,并支持跨组织进行更有意义的比较。
For organizations seeking to use the QCAT outputs to drive targeted improvements across maturity levels, establishing a clear baseline of current maturity across each element should be assessed before initiating change management interventions to monitor for effectiveness. The QCAT framework provides explicit descriptions of each maturity level from behavioral and operational characteristics, which enables organizations to identify gaps and systematically pursue improvements. This staged approach supports structured elevation of quality culture rather than isolated element score optimization, and reinforces a holistic, systems-based view of quality culture development.
对于寻求使用QCAT输出来推动跨成熟度水平针对性改进的组织,在启动变革管理干预措施以监测有效性之前,应评估各要素当前成熟度的明确基线。QCAT框架从行为和运营特征方面提供了每个成熟度水平的明确描述,使组织能够识别差距并系统地追求改进。这种分阶段方法支持质量文化的结构化提升而非孤立的要素评分优化,并强化了对质量文化发展的整体性、基于系统的视角。
This study reinforces the importance of adopting a data-driven approach to pharmaceutical QMM, which acknowledges both behavioral and structural influences on quality culture. The variability in the dataset observed across sites suggests that rating outcomes are shaped by a complex interplay of organizational design, leadership behavior, and operational priorities, rather than compliance status or resource availability alone.
本研究强化了采用数据驱动方法进行药品QMM的重要性,该方法承认对质量文化的行为和结构影响。跨场地观察到的数据集变异性表明,评分结果由组织设计、领导行为和运营优先事项的复杂相互作用所塑造,而非仅由合规状态或资源可用性决定。
Further analysis and refinement of the QCAT with a larger and more heterogeneous dataset are warranted, particularly for the Leadership Commitment construct, which showed the weakest internal consistency, inter-item coherence, and structural fit. Expanding the sample to include smaller manufacturers and linking scores to downstream quality or patient outcomes would enhance generalizability. Future work may also leverage leadership maturity models from other industries to refine this domain.
有必要使用更大且更多样化的数据集对QCAT进行进一步分析和完善,特别是对于显示出最弱内部一致性、项目间连贯性和结构拟合的领导承诺构念。扩大样本以纳入小型生产商并将评分与下游质量或患者结果相联系将增强普适性。未来工作还可利用其他行业的领导成熟度模型来完善这一领域。
Limitations
局限性
While several domains yielded statistically significant or marginal omnibus differences across site region and business function, very few pairwise post hoc comparisons reached significance after correcting for multiple testing. This pattern suggests limited discriminative power at the item level and raises questions about the tool’s ability to meaningfully differentiate between group-level responses in its current form. Although this could reflect true cultural convergence across the industry, driven by regulatory compliance and the critical nature of patient safety, it may also stem from restricted variability or ceiling effects in certain domains. In particular, three of the four Leadership Commitment elements (e.g., Safety Program: median=4.0, skew=−1.16; Rewards and Recognition: median=3.0, skew=−0.82; and Feedback and Staff Development: median=2.0, skew=1.04) show distributional patterns consistent with compression at the upper or lower ends of the scale (see Supplement 2). These features may have reduced the tool’s sensitivity to detect between-group differences, even where genuine organizational variation exists.
虽然若干领域在场地区域和业务功能间产生了统计显著或边缘的总差异,但校正多重检验后很少有事后两两比较达到显著性。这一模式表明项目水平的区分能力有限,并引发关于该工具在当前形式下有意义地区分组间响应能力的质疑。虽然这可能反映了行业间真正的文化趋同——由监管合规和患者安全的关键性质所驱动——但也可能源于某些领域的受限变异或天花板效应。特别是,四个领导承诺要素中的三个(如安全计划:中位数=4.0,偏度=-1.16;奖励与认可:中位数=3.0,偏度=-0.82;反馈与员工发展:中位数=2.0,偏度=1.04)显示出与量表上端或下端压缩一致的分布模式(见补充材料2)。这些特征可能降低了工具检测组间差异的敏感性,即使存在真正的组织变异。
Given its lower internal consistency (Cronbach’s α=0.49), the Leadership Commitment category warrants cautious interpretation in this study. Safety Program and Rewards and Recognition elements currently grouped under the Leadership Commitment category may not be directly influenced by site-level quality leadership, and instead reflect broader organizational structures (e.g., EHS or HR). This raises the possibility that the Leadership Commitment construct, as currently framed, may conflate distinct dimensions of organizational culture.
鉴于其较低的内部一致性(克朗巴赫系数α=0.49),本研究中的领导承诺类别需要谨慎解释。目前归入领导承诺类别的安全计划和奖励与认可要素可能不受场地级质量领导的直接影响,而是反映更广泛的组织结构(如EHS或HR)。这引发了领导承诺构念在当前框架下可能混淆组织文化不同维度的可能性。
The dataset also reflects organizations that voluntarily participated and had invested in staff trained on PDA’s QCAT, likely a selection bias toward companies with mature quality systems. This bias may have influenced the observed scores in the original 58 datapoints. Future studies must include a more diverse range of companies and facilities.
该数据集还反映了自愿参与并投资于PDA QCAT员工培训的组织的特征,这可能存在对拥有成熟质量体系的公司的选择偏倚。这种偏倚可能影响了原始58个数据点中观察到的评分。未来研究必须纳入更多样化的公司和场地范围。
The overall consistency of findings across multiple organizational types reinforces the robustness of the PDA’s original category framework. The alignment between most measured elements and their respective categories illustrates that the PDA’s quality culture domains provide a solid and empirically supported foundation for assessing organizational maturity, particularly for the future of the QMM paradigm.
跨多种组织类型发现的整体一致性强化了PDA原始类别框架的稳健性。大多数测量要素与其相应类别之间的对齐表明,PDA的质量文化领域为评估组织成熟度提供了坚实且经验支持的基础,特别是对于QMM范式的未来。
Conclusion
结论
This study provides empirical support for the PDA QCAT Framework as a valid and relevant instrument for assessing organizational quality cultural maturity in the pharmaceutical industry. Variability across business functions and product types underlies the need for contextual benchmarking and flexible evaluation within the QMM framework. This work supports the data-driven, adaptable approach to QMM that promotes continuous improvement, regulatory compliance, and sustained product quality in the evolution to achieving product realization to patients.
本研究为PDA QCAT框架作为评估制药行业组织质量文化成熟度的有效且相关工具提供了经验支持。业务功能和产品类型间的变异性凸显了在QMM框架内进行情境化基准测试和灵活评估的必要性。这项工作支持了QMM的数据驱动、适应性方法,该方法促进持续改进、监管合规和持续产品质量,以实现向患者提供产品的目标。
Supplementary Information The online version contains supplementary material available at https://doi.org/10.1208/s12248-026-01234-x.
补充信息 在线版本包含可获得的补充材料,网址:https://doi.org/10.1208/s12248-026-01234-x。
Acknowledgements We sincerely thank Tita Tavares and David Tal-madge of the Parenteral Drug Association (PDA) for providing the data. Additionally, we acknowledge the contributions of the students (Claire Manning, Siena Samione, Ava Pandey, Dylan Krueger, Claudia Gibson, and Aleigh Diaz) and faculty (William Raasch) at Keck Graduate Institute (KGI) for their support and engagement throughout this project.
致谢 我们衷心感谢注射剂协会(PDA)的Tita Tavares和David Tal-madge提供数据。此外,我们感谢Keck研究生院(KGI)的学生(Claire Manning、Siena Samione、Ava Pandey、Dylan Krueger、Claudia Gibson和Aleigh Diaz)和教师(William Raasch)在整个项目中的支持和参与。
Funding Open access funding provided by SCELC, Statewide Califor-nia Electronic Library Consortium. Institutional support was provided by Keck Graduate Institute.
资助 开放获取资金由SCELC(全州加州电子图书馆联盟)提供。机构支持由Keck研究生院提供。
Data Availability The PDA data used in this study was obtained from the Parenteral Drug Association (PDA) through their Culture Assessment Tool. Access to the data is subject to PDA’s data sharing policies. Interested researchers may contact PDA directly to inquire about data access and further information on collaboration opportunities.
数据可用性 本研究使用的PDA数据通过注射剂协会(PDA)的文化评估工具获得。数据访问受PDA数据共享政策约束。有兴趣的研究人员可直接联系PDA询问数据访问和合作机会的进一步信息。
Declarations
声明
Conflict of Interest The authors declare no competing interests.
利益冲突 作者声明无竞争利益。
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
开放获取 本文采用知识共享署名4.0国际许可协议授权,允许在任何媒介或格式中使用、共享、改编、分发和复制,只要您对原作者和来源给予适当署名,提供知识共享许可协议的链接,并指明是否进行了更改。本文中的图片或其他第三方材料包含在文章的知识共享许可协议中,除非在材料来源行中另有说明。如果材料未包含在文章的知识共享许可协议中,且您的预期使用不受法定法规允许或超出允许使用范围,您将需要直接从版权持有者获得许可。要查看该许可协议的副本,请访问http://creativecommons.org/licenses/by/4.0/。
References
参考文献
-
FDA. Drug shortages: Root causes and potential solutions [Internet]. 2020. Retrieved 7 May 2025; from: https://www.fda.gov/drugs/drug-shortages/report-drug-shortages-root-causes-and-potential-solutions
FDA. 药品短缺:根本原因和潜在解决方案[互联网]. 2020. 2025年5月7日检索;来自:https://www.fda.gov/drugs/drug-shortages/report-drug-shortages-root-causes-and-potential-solutions
-
Friedman R. Establishing a culture of quality [Internet]. 2021; Silver Spring, MD. Retrieved 7 May 2025; from: https://www.fda.gov/media/156357/download
Friedman R. 建立质量文化[互联网]. 2021;马里兰州银泉市. 2025年5月7日检索;来自:https://www.fda.gov/media/156357/download
-
Harouaka A. 2023 FDA QMM Overview [Internet]. 2023. Retrieved 7 May 2025; from: https://www.fda.gov/media/169940/download
Harouaka A. 2023 FDA QMM概述[互联网]. 2023. 2025年5月7日检索;来自:https://www.fda.gov/media/169940/download
-
Rakala, Nandini. Quality Management Maturity (QMM) [Internet]. Pharmaceutical quality symposium 2023: Quality, supply chain & advanced manufacturing; 2023. Retrieved 7 May 2025; from: https://www.fda.gov/media/174243/download
Rakala, Nandini. 质量管理成熟度(QMM)[互联网]. 2023年药品质量研讨会:质量、供应链与先进制造;2023. 2025年5月7日检索;来自:https://www.fda.gov/media/174243/download
-
FDA. Quality metrics for drug manufacturing [Internet]. FDA; 2024. Retrieved 7 May 2025; from: https://www.fda.gov/drugs/pharmaceutical-quality-resources/quality-metrics-drug-manufacturing
FDA. 药品生产质量指标[互联网]. FDA;2024. 2025年5月7日检索;来自:https://www.fda.gov/drugs/pharmaceutical-quality-resources/quality-metrics-drug-manufacturing
-
FDA. Food and Drug Administration Quality Metrics Reporting Program; Establishment of a Public Docket; Request for Comments [Internet]. 2022. Retrieved 7 May 2025; from: https://www.federalregister.gov/documents/2022/03/09/2022-04972/food-and-drug-administration-quality-metrics-reporting-program-establishment-of-a-public-docket
FDA. 食品药品监督管理局质量指标报告计划;建立公共案卷;征求意见[互联网]. 2022. 2025年5月7日检索;来自:https://www.federalregister.gov/documents/2022/03/09/2022-04972/food-and-drug-administration-quality-metrics-reporting-program-establishment-of-a-public-docket
-
Parenteral Drug Association. Comments on FDA Quality Metrics Reporting Program [Internet]. 2022. Retrieved 7 May 2025; from: https://www.regulations.gov/comment/FDA-2022-N-0075-0021
注射剂协会. 关于FDA质量指标报告计划的评论[互联网]. 2022. 2025年5月7日检索;来自:https://www.regulations.gov/comment/FDA-2022-N-0075-0021
-
AstraZeneca. Comments on FDA quality metrics reporting program [Internet]. 2022. Retrieved 7 May 2025; from: https://www.regulations.gov/comment/FDA-2022-N-0075-0077
阿斯利康. 关于FDA质量指标报告计划的评论[互联网]. 2022. 2025年5月7日检索;来自:https://www.regulations.gov/comment/FDA-2022-N-0075-0077
-
Association of Accessible Medicines. AAM quality metrics white paper [Internet]. 2021. Retrieved 7 May 2025; from: https://www.aam adwshus.net/wp-content/uploads/2024/7_Michaill-at-el.-1.pdf
可及药物协会. AAM质量指标白皮书[互联网]. 2021. 2025年5月7日检索;来自:https://www.aam adwshus.net/wp-content/uploads/2024/7_Michaill-at-el.-1.pdf
-
Maguire J, Fisher A, Harouaka D, Rakala N, Lundi C, Yambot M, et al. Lessons from CDER’s quality management maturity program [Internet]. 2023; 23:25:14. https://doi.org/10.1208/ s12248-022-00777-z
Maguire J, Fisher A, Harouaka D, Rakala N, Lundi C, Yambot M等. CDER质量管理成熟度计划的经验教训[互联网]. 2023;23:25:14. https://doi.org/10.1208/s12248-022-00777-z
-
FDA. Quality management initiatives in the pharmaceutical industry: an economic perspective [Internet]. 2025. Retrieved 7 May 2025; from: https://www.fda.gov/media/187861/download?attach ment
FDA. 制药行业质量管理倡议:经济视角[互联网]. 2025. 2025年5月7日检索;来自:https://www.fda.gov/media/187861/download?attachment
-
FDA. Quality management maturity: essential for stable U.S. supply chains of quality pharmaceuticals [Internet]. 2022. Retrieved 7 May 2025; from: https://www.fda.gov/media/157432/download
FDA. 质量管理成熟度:稳定美国优质药品供应链的关键[互联网]. 2022. 2025年5月7日检索;来自:https://www.fda.gov/media/157432/download
-
FDA. CDER’s quality management maturity (QMM) program: practice areas and prototype assessment protocol development [Internet]. Silver Spring, MD; 2023. Retrieved 7 May 2025; from: https://www.fda.gov/media/171705/download
FDA. CDER质量管理成熟度(QMM)计划:实践领域和原型评估协议开发[互联网]. 马里兰州银泉市;2023. 2025年5月7日检索;来自:https://www.fda.gov/media/171705/download
-
Michalik R, Hauck W. The FDA’s quality management maturity program: an overview [Internet]. 2024. Retrieved 7 May 2025; from: https://www.news-and-articles/NewsArticles/2024/7/The-FDA-s-Quali-ty-Management-Maturity-Program
Michalik R, Hauck W. FDA质量管理成熟度计划概述[互联网]. 2024. 2025年5月7日检索;来自:https://www.news-and-articles/NewsArticles/2024/7/The-FDA-s-Quali-ty-Management-Maturity-Program
-
FDA. Voluntary quality management maturity prototype assessment protocol evaluation program [Internet]. 2024. Retrieved 7 May 2025; from: https://www.federalregister.gov/documents/2024/05/07/2024-05205/voluntary-quality-management-maturity-prototype-assessment-protocol-evaluation-program
FDA. 自愿性质量管理成熟度原型评估协议评估计划[互联网]. 2024. 2025年5月7日检索;来自:https://www.federalregister.gov/documents/2024/05/07/2024-05205/voluntary-quality-management-maturity-prototype-assessment-protocol-evaluation-program
-
Pradit T, Bues B, Köhler S, Chen C, Mendivil S, Baker D. The impact of quality culture on operational performance—An empirical study from the pharmaceutical industry. PDA J Pharm Sci Technol. 2018;72(5):531–542. Available from: http://doi.org/10. 5731/pdajpst.2018.00871
Pradit T, Bues B, Köhler S, Chen C, Mendivil S, Baker D. 质量文化对运营绩效的影响——来自制药行业的实证研究. PDA J Pharm Sci Technol. 2018;72(5):531-542. 可获取于:http://doi.org/10.5731/pdajpst.2018.00871
-
Patel P, Baker D, Burdick R, Chen C, Hill J, Holland M, et al. Quality culture: a PDA perspective. PDA J Pharm Sci Technol. 2015;69(5):631–42. Available from: https://journal.pda.org/conte nt/69/5/631
Patel P, Baker D, Burdick R, Chen C, Hill J, Holland M等. 质量文化:PDA视角. PDA J Pharm Sci Technol. 2015;69(5):631-42. 可获取于:https://journal.pda.org/content/69/5/631
-
Fellows M, Friedli T, Li Y, Maguire J, Rakala N, Ritz M, et al. Benchmarking the quality practices of global pharmaceutical manufacturing to advance supply chain resilience. AAPS J [Internet]. 2022;24:111. https://doi.org/10.1208%2Fs12248-022-00761-7.
Fellows M, Friedli T, Li Y, Maguire J, Rakala N, Ritz M等. 全球药品生产质量实践基准测试以促进供应链韧性. AAPS J [互联网]. 2022;24:111. https://doi.org/10.1208%2Fs12248-022-00761-7.
 夜雨聆风
夜雨聆风




