文档内容
学科网(北京)股份有限公司
龙岩市一级校2022-2023 学年第一学期期末高三联考
化学参考答案
一、选择题:本题共10 小题,每小题2 分,共20 分。
题号
1
2
3
4
5
6
7
8
9
10
答案
C
B
D
B
A
A
C
C
A
D
二、非选择题:本题共5 小题,共60 分。
11.(12 分)
(1)多次浸取;适当增大稀硫酸浓度;充分搅拌;适当升高温度;粉碎(任答1 种,其他合理答案也给分)(1 分)
(2)Al(OH)3............................................................................................................................ (1 分)
3Fe2++MnO -
4
+8H2O === MnO(OH)2↓+3Fe(OH)3↓+5H+....................................(2 分)
(3)Zn......................................................................................................................................(1 分)
(4)5Zn2++10HCO3
- === Zn5(CO3)2(OH)6·2H2O↓+8CO2↑............................................(2 分)
(5)n[Zn(OH)2]∶n(ZnO)=1∶4............................................................................................ (2 分)
(6)加入最后一滴氢氧化钠溶液,溶液颜色由无色变为浅红色且半分钟内不变色..... (1 分)
97.2%............................................................................................................................... (2 分)
12.(11 分)
(1)Cu+4HNO3 === Cu(NO3)2+2NO2↑+2H2O................................................................ (2 分)
(2)HNO3................................................................................................................................ (1 分)
(3)Cu(NO3)2.......................................................................................................................... (1 分)
向④中溶液通入NO2 气体后,加入铜片..................................................................... (1 分)
N2(或稀有气体等其他不与体系发生反应的气体)..................................................(1 分)
HNO2.................................................................................................................................(1 分)
(4)排除通NO2 带来的硝酸浓度增大的影响.....................................................................(1 分)
(5)2HNO3+H2O2+Cu === Cu(NO3)2+2H2O...................................................................(1 分)
90%.................................................................................................................................. (2 分)
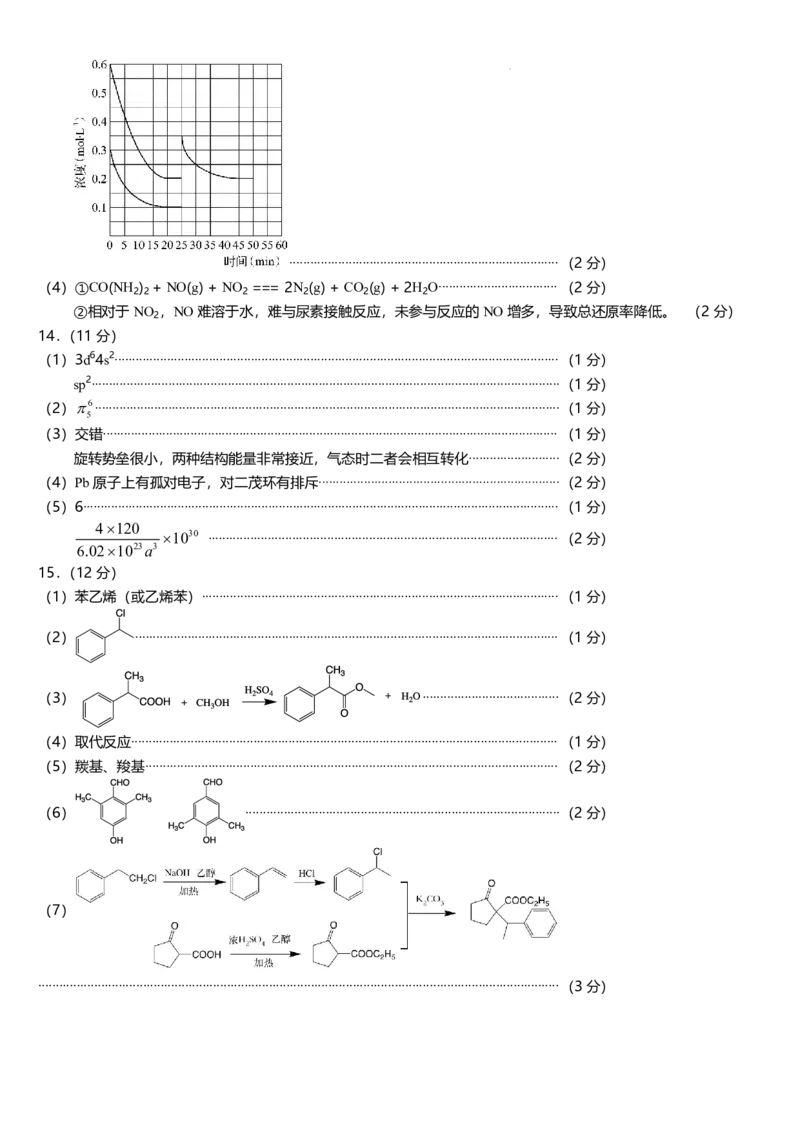
13.(14 分)
(1)2NH3(g)+CO2(g)
CO(NH2)2(s)+H2O(g) ,△H1= - 87.0 kJ/mol....................... (2 分)
(2)该反应正向放热,气体分子数减小,高压低温有利于平衡右移,提高原料利用率。
(2 分)
(3)①ABD..............................................................................................................................(2 分)
②44.4%........................................................................................................................... (1 分)
50..................................................................................................................................(1 分)
③
学科网(北京)股份有限公司
............................................................................. (2 分)
(4)①CO(NH2)2 + NO(g) + NO2 === 2N2(g) + CO2(g) + 2H2O.................................. (2 分)
②相对于NO2,NO 难溶于水,难与尿素接触反应,未参与反应的NO 增多,导致总还原率降低。
(2 分)
14.(11 分)
(1)3d64s2............................................................................................................................... (1 分)
sp2......................................................................................................................................(1 分)
(2)
6
5
.....................................................................................................................................(1 分)
(3)交错.................................................................................................................................. (1 分)
旋转势垒很小,两种结构能量非常接近,气态时二者会相互转化..........................(2 分)
(4)Pb 原子上有孤对电子,对二茂环有排斥.....................................................................(2 分)
(5)6........................................................................................................................................ (1 分)
30
23
3
4 120
10
6.02 10 a
.................................................................................................... (2 分)
15.(12 分)
(1)苯乙烯(或乙烯苯)...................................................................................................... (1 分)
(2)
......................................................................................................................... (1 分)
(3)
.......................................(2 分)
(4)取代反应.......................................................................................................................... (1 分)
(5)羰基、羧基...................................................................................................................... (2 分)
(6)
..........................................................................................(2 分)
(7)
.....................................................................................................................................................(3 分)